This is an interesting/informative post on B12 vitamins from Metagenics/Dr Mercola’s :
In a broad sense B-12 still refers to a group of cobalt-containing vitamer compounds known as cobalamins:
these include
1)cyanocobalamin (an artifact formed as a result of the use of cyanide in the purification procedures),
2)hydroxocobalamin (natural precurser form),
and finally, the two naturally occurring cofactor forms of B-12:
3) 5-deoxyadenosylcoba lamin (adenosylcobalaminâ”AdoB-12), the cofactor of Methylmalonyl Coenzyme A mutase (MUT),
4)Methylcobalamin (MeB-12), the cofactor of 5-methyltetrahydrof olate-homocysteine methyltransferase (MTR).
Methylcobalamin supporters state its superiority as it is the primary circulatory form and acts as a methyl donor. Adenosylcobalamin, however, accounts for 70% of cobalamin stored in the livert the major storage site for B12, while methylcobalamin accounts for only 1% to 3%. It is also argued that cyanocobalamin is poorly converted into its active forms and releases cyanide into circulation-neither of these statements is supported by research.
Since the cyanocobalamin form of B-12 is deeply red colored, easy to crystallize, and is not sensitive to air-oxidation, it is typically used as a form of B-12 for food additives and in many common multivitamins. Research findings have demonstrated that oral cyanocobalamin is safe and easily and rapidly converted to both methylcobalamin and adenosylcobalamin during absorption and at the target cell, and does reverse B12 deficiency signs and symptoms.
Oral cyanocobalamin has a long history of use worldwide. In Sweden, oral high-dose cyanocobalamin is the major treatment form for B12 deficiency and maintenance and has gained widespread popularity since its introduction in 1964. More than one million patients and years of data in Sweden support the use of oral cyanocobalamin to correct and prevent B12 deficiency signs and symptoms and it is considered a standard of care for most patients. In virtually every aspect of B12 activity oral cyanocobalamin has demonstrated benefits, including psychological, neurological, and hematological.
A key function of B12 is its participation in methylation reactions. Foremost of these processes is the reduction homhomocysteine.
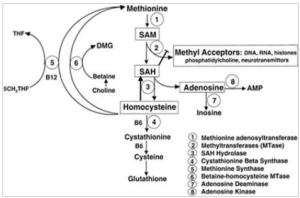
While methylcobalamin does participate in homocysteine reduction, it is secondary in significance to folic acid. Methylcobalamin receives its methyl group from folic acid (methyltetrahydrofo late). The body ârecyclesâ methyl groups and cobalamin. In the homocysteine cycle, as an example, cobalamin donates its methyl group and is then converted back to methylcobalamin, receiving a methyl group from 5-methyl-tetrahydro folate. The primary methyl donor is folic acid along with other methyl donors, whereas cyanocobalamin provides the vitamin B12 component for the cycle. Â In patients with end stage renal disease, a condition associated with hyperhomocysteinemia cyanocobalamin was demonstrated to be equipotent in reducing plasma homocysteine levels in a comparison to hydroxycobalamin.
In a 2001 study printed in JAMA, Tice et al. reported oral cyanocobalamin to be a cost-effective method of reducing plasma homocysteine levels in multiple population groups. Â B12 deficiency has been associated with alterations in cognition in the elderly. There is a known connection between elevations in homocysteine and age-related cognitive decline. The relationship must certainly include deficiencies of both folic acid and B12.
Oral cyanocobalamin is capable of reducing serum methylmalonic acid concentrations, an indication of B12 repletion. Therefore, increasing the intake of B12 as cyanocobalamin may provide protection against cognitive decline in older populations.
No toxic effects of oral B12 consumption have ever been reported at any level of intake. In a 1991 JAMA report, Hatchcock and Troendle reported no concerns with the oral use of B12. Cyanide release from oral B12 was said to be toxicologically insignificant. The lack of reported B12 toxicity is a testament to the effective and safe use of this oral compound.
http://en.wikipedia .org/wiki/ Vitamin_B12
In humans, only two corresponding coenzyme B-12-dependent enzymes are known:
-Methylmalonyl Coenzyme A mutase (MUT) which uses the AdoB-12 form and reaction type 1 to catalyze a carbon skeleton rearrangement (the X group is -COSCoA). MUT’s reaction converts MMl-CoA to Su-CoA, an important step in the extraction of energy from proteins and fats. This functionality is lost in vitamin B-12 deficiency, and can be measured clinically as an increased methylmalonic acid (MMA) level. Unfortunately, an elevated MMA, though sensitive to B-12 deficiency, is probably overly sensitive, and not all who have it actually have B-12 deficiency. For example, MMA is elevated in 90-98% of patients with B-12 deficiency; however 25-20% of patients over the age of 70 have elevated levels of MMA, yet 25-33% of them do not have B-12 deficiency. For this reason, MMA is not routinely recommended in the elderly.[11] The “gold standard” test for B-12 deficiency continues to be low blood levels of the vitamin. The MUT function cannot be affected by folate supplementation, and which is necessary for myelin synthesis (see mechanism below) and certain other functions of the central nervous system.
Other functions of B-12 related to DNA synthesis related to MTR dysfunction (see below) can often be corrected with supplementation with the vitamin folic acid, but not the elevated levels of homocysteine, which is normally converted to methionine by MTR.
5-methyltetrahydrofolate-homocystei ne methyltransferase (MTR), also known as methionine synthase. This is a methyl transfer enzyme, which uses the MeB-12 and reaction type 2 to catalyze the conversion of the amino acid Hcy back into Met.[12] This functionality is lost in vitamin B-12 deficiency, and can be measured clinically as an increased homocysteine level in vitro. Increased homocysteine can also be caused by a folic acid deficiency, since B-12 helps to regenerate the tetrahydrofolate (THF) active form of folic acid. Without B-12, folate is trapped as 5-methyl-folate, from which THF cannot be recovered unless a MTR process reacts the 5-methyl-folate with homocysteine to produce methionine and THF, thus decreasing the need for fresh sources of THF from the diet. THF may be produced in the conversion of homocysteine to methionine, or may be obtained in the diet.
It is converted by a non-B-12-dependent process to 5,10-methylene- THF, which is involved in the synthesis of thymine. Reduced availability of 5,10-methylene- THF results in problems with DNA synthesis, and ultimately in ineffective production cells with rapid turnover, in particular blood cells, and also intestinal wall cells which are responsible for absorption. The failure of blood cell production results in the once-dreaded and fatal disease, pernicious anemia. All of the DNA synthetic effects, including the megaloblastic anemia of pernicious anemia, resolve if sufficient folate is present (since levels of 5,10-methylene- THF still remain adequate with enough dietary folate). Thus the best known function of B-12 (that which is indirectly involved with DNA synthesis and restoration of cell-division and anemia) is actually a facultative function which is mediated by B-12 conservation of active folate which can be used for DNA production.










